Solutions in which water is the solvent are, of course, very common on our planet. This component is called the solvent and may be viewed as the medium in which the other components are dispersed, or dissolved. Often, though not always, a solution contains one component with a concentration that is significantly greater than that of all other components. The relative amount of a given solution component is known as its concentration. We will explore a more thorough treatment of solution properties in the chapter on solutions and colloids, but here we will introduce some of the basic properties of solutions. Solutions occur frequently in nature and have also been implemented in many forms of manmade technology.

We have previously defined solutions as homogeneous mixtures, meaning that the composition of the mixture (and therefore its properties) is uniform throughout its entire volume. The amount of sugar in a given amount of coffee is an important determinant of the beverage’s sweetness. In this section, we will describe one of the most common ways in which the relative compositions of mixtures may be quantified.įigure 3.14 Sugar is one of many components in the complex mixture known as coffee. The relative amount of sugar in a beverage determines its sweetness (see Figure 3.14). The relative amount of the active ingredient in a medicine determines its effectiveness in achieving the desired pharmacological effect. The relative amounts of iron, carbon, nickel, and other elements in steel (a mixture known as an “alloy”) determine its physical strength and resistance to corrosion. The relative amount of oxygen in a planet’s atmosphere determines its ability to sustain aerobic life. Similar to a pure substance, the relative composition of a mixture plays an important role in determining its properties. However, mixtures-samples of matter containing two or more substances physically combined-are more commonly encountered in nature than are pure substances. In preceding sections, we focused on the composition of substances: samples of matter that contain only one type of element or compound. Perform dilution calculations using the dilution equation.Calculate solution concentrations using molarity.Describe the fundamental properties of solutions.Molar mass is measured in grams per mole.Learning Objectives By the end of this section, you will be able to: The mass in grams of one mole of a material is known as molar mass. What is molar mass, and what is its unit? The mass of a specimen of a chemical compound divided by the amount of material in that specimen, expressed in moles, is the molar mass of that compound.ģ. Add everything up and convert to grams/mole.Multiply every element’s atomic weight by the number of atoms included in the compound.Calculate the number of atoms of every element in the compound using the chemical formula.The standard unit is g mol⁻¹ and the SI one as kg mol⁻¹.Molar mass can be defined as the smallest unit of mass of any compound and is the twelfth of one carbon mass – 12 atoms.Find the number of moles in a 39.25-gram sample of CdBr₂.Calculate the molar mass of KAl(SO₄)₂♱2 H₂O.Here are some questions for you to practise: Let’s take another example to find the mass of sulfur in an 8.5 mol sample of sulfur: To get the final answer, add up the total values, i.e., 46 12 48 = 106Hence, the molar mass of Na₂CO₃ is 106 g/mole. Now that you know the atomic mass and how many atoms of the compound are involved, you can find the molar mass as shown below. Count the number of atoms present in each element (The subscript of an element in its chemical formula).

For any element, the number that appears on the top left represents atomic mass. For each element, you must figure out the atomic mass using the periodic table.
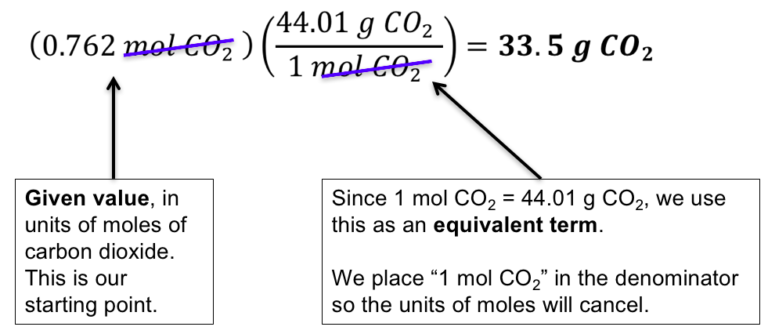
Source HOW TO CALCULATE THE MOLAR MASS OF A COMPOUND? In any substance, the number of entities present can always be defined as a mole. The molar mass of any substance is defined as the smallest mass unit of that compound with one-twelfth of the mass of one carbon that would be 12 atoms. So, how do you estimate the molar mass mathematically? WHAT IS MOLAR MASS? The molar mass of a compound is computed by summing the atomic masses of all the atoms in the molecule. The molar mass is the mass of one sampling mole. Have you ever wondered how many air molecules there are in the soccer ball you’re playing with or the food you’re eating? Atoms or molecules are the smallest units in terms of mass and size, as we all understand.
